What is the UK Conformity Assessed Mark (UKCA)?
The United Kingdom Conformity Assessed (UKCA) mark is replacing the CE mark in Great Britain, as the United Kingdom left the EU single market on 31 December 2020. It is being introduced in a phased approach, with the CE mark remaining acceptable when placing most products on the GB market until 31 December 2022. However, for Medical Devices, the deadline is June 30th 2023
What is the UKCA mark?
According to the UK Government, the UKCA mark is “a new product marking that is being used for goods being placed on the market in Great Britain (England, Wales and Scotland). It covers most goods which previously required the CE marking known as “new approach” goods.”
It is important to note that the UKCA mark cannot be used for goods in the Northern Ireland market. These goods will still require CE marking for self-assessed goods and those that are conformity assessed by an EU Notified Body. The CE plus UKNI mark is used for goods that are conformity assessed by a UK Notified Body.
The purpose of the UKCA mark is to indicate that the product it is attached to conforms to GB legislation. The manufacturer, or their authorised representative, is responsible for affixing the UKCA mark to their product.
When does the UKCA mark come into force?
The UKCA marking came into effect on 1 January 2021. However, in most cases, CE marking is still acceptable until 1 January 2023 to allow businesses time to adjust.
After 1 January 2021, only products with UKCA marking will be accepted in Great Britain.
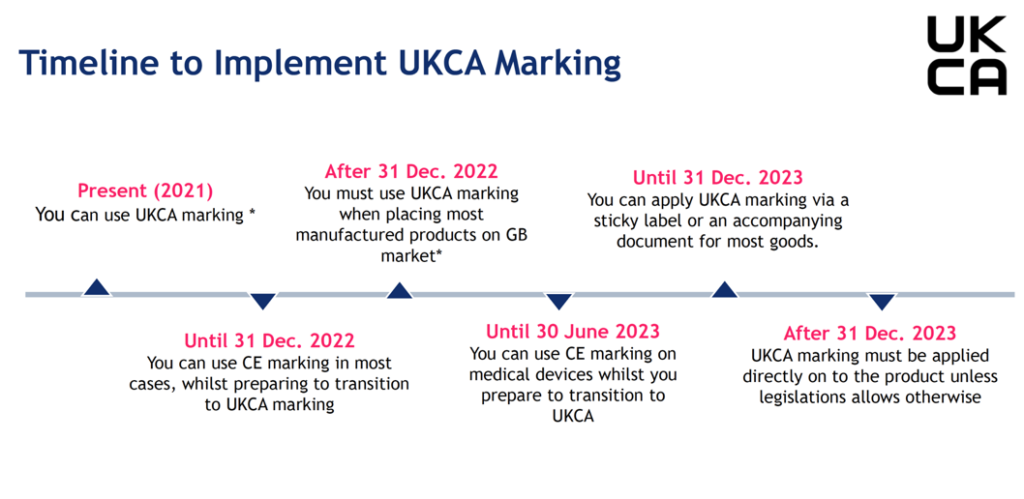
The end date for recognising the CE mark in Great Britain will differ for certain goods, such as medical devices.
How does the UKCA mark affect medical device manufacturers?
#1 If you are a UK-based manufacturer supplying to the UK market only
- If it is not already registered, you need to register your device with the MHRA (Medicines and Healthcare Products Regulatory Agency).
- MDD (Medical Devices Directive) and MHRA certification will be accepted in Great Britain until 30 June 2023. The UKCA mark will be necessary after this date, and MDR will not be mandatory.
- If you are based in Northern Ireland, the CE mark is required, and MDR has been mandatory from 26 May 2021. Valid MDD certificates will be accepted until their expiry date (and no later than 25 May 2024) as long as there are no substantial changes to the device.
#2 If you are a UK-based manufacturer supplying the EU
- Since 1 January 2021, you must have had an EU or Ni-based authorised representative.
- You must also comply with the CE mark (MDD/MDR).
#3 If you are outside the UK and supplying to the UK market
- If you supply to Great Britain, the CE mark will be recognised until 30 June 2023, with the UKCA mark required after this. A dual certification, CE + UKCA, will also be accepted.
- The CE mark and MDR certification will also be required if you supply to the Northern Ireland (NI) market. MDD-certified devices can be placed on the NI market until 24 May 2024 (unless there are substantial changes to the device, which will require MDR certification).
- You will also have to register with a UK-based responsible person.
Why work with Shiro Business Solutions?
Shiro Business Solutions is highly experienced in ensuring medical devices and medical technology companies are operating as efficiently as possible and are compliant with the rapidly changing regulatory scenarios of the EU. MDR and IVDR.
Please contact us to get a jargon-free service regarding your UKCA requirements.

